Subtle risks make headlines, but toxicology and epidemiology don’t support the autism claim.
Why I’m Writing This (with some help)
A long time ago, I worked as a product regulatory specialist in the petrochemical industry.
The title sounds grand, but in practice my role was straightforward: authoring and managing Safety Data Sheets (SDS). That experience—part mentorship, part self-teaching, part hard-earned practice—still shapes how I think about chemical safety today.
Whenever I see headlines screaming that “food dyes are killing you” or “painkillers cause autism,” my instinct is to go back to ground zero: the SDS, particularly Section 11—Toxicological Data.
Now, I don’t claim deep clinical insight into medical outcomes. But you can learn a lot just by reading the chemistry. Two of the key flags in toxicology—mutagenicity (the potential of a substance to cause permanent, heritable changes in DNA) and teratogenicity (the ability to cause congenital malformations or developmental abnormalities in a fetus)—are not considered risk factors for acetaminophen.
Why spell this out? Because while public figures like RFK Jr. wield outsized influence in shaping public fears around health, it’s essential to fight misinformation (and disinformation) with information. That doesn’t mean dumbing it down; it means getting into the weeds and showing people how to parse the evidence for themselves.
That’s the lens I bring to the current controversy linking Tylenol and autism—one grounded less in clickbait headlines and more in the unglamorous but reliable language of toxicology.
Section 11: Toxicological Information
If you’ve never seen one before, an SDS (Safety Data Sheet) may look like dry paperwork meant for compliance officers and lab techs. But it’s the gold standard reference when assessing chemical risks. Each section has a purpose, and Section 11—Toxicological Information—is where the hard data lives. This is the part that cuts through headline noise, laying out what is actually known about a substance’s acute and chronic effects, its potential for mutagenicity, carcinogenicity, or reproductive harm.
In other words, Section 11 is the sober counterweight to the panic of “chemicals cause X.” By showing the toxicology in plain numbers and classifications, it becomes possible to separate genuine hazard from speculative fear. With that in mind, here’s what Section 11 tells us about acetaminophen (paracetamol).
Section 11: Toxicological Information
This section provides toxicological data for the pure substance, which is critical for risk assessment, especially in occupational settings. The clinical effects of overdose are well-documented in medical literature.
11.1. Information on Toxicological Effects
Acute Toxicity:
Oral (LD<sub>50</sub>): Approximately 2,000 - 4,000 mg/kg (rat). This value is misleading for human risk assessment. Human toxicity is not directly comparable to rodent data. In humans, acute oral toxicity is characterized by a dose-dependent threshold.
Doses exceeding 150 mg/kg (or 7.5 - 10 g total for an adult) are considered potentially toxic and require medical evaluation.
Doses exceeding 250 mg/kg carry a high risk of severe, potentially fatal, hepatotoxicity.
Dermal (LD<sub>50</sub>): Not applicable. Expected to be low via this route due to poor skin absorption.
Inhalation (LC<sub>50</sub>): Data not available. Not expected to be a significant route of acute toxicity for the solid powder, but dust may cause respiratory irritation.
Skin Corrosion/Irritation: Not classified. Not expected to be irritating to skin.
Serious Eye Damage/Irritation: Not classified. May cause mechanical irritation as a solid powder (dust), but not chemically irritating.
Respiratory or Skin Sensitization: Not classified. Evidence of sensitization is rare.
Germ Cell Mutagenicity: Negative results in multiple in vitro and in vivo tests (including Ames test). Not considered mutagenic.
Teratogenicity: Not classified as a reproductive toxicant under the GHS criteria specific for teratogenicity (causing structural birth defects).
Data: Based on extensive human epidemiological data, the substance is not associated with a significant increase in major congenital malformations when used at therapeutic doses during pregnancy.
Carcinogenicity:
IARC: Not classified as a carcinogen to humans (Group 3).
NTP: Not listed as a carcinogen.
ACGIH: Not classified as a carcinogen.
Note: Some studies on high-dose, long-term exposure have shown ambiguous results, but no conclusive evidence links therapeutic doses to cancer in humans.
Reproductive Toxicity: Not classified for specific reproductive effects. Therapeutic doses are not considered to impair fertility. It is commonly used during pregnancy but should only be done so under medical advice.
STOT - Single Exposure (Specific Target Organ Toxicity): Category 1 (Liver)
Hazard Class: Hepatotoxicity.
Route of Exposure: Ingestion.
Effects: Single exposure through overdose can cause acute liver failure, centrilobular hepatic necrosis, and is a leading cause of acute liver failure in many countries.
STOT - Repeated Exposure (Specific Target Organ Toxicity): Category 1 (Liver)
Hazard Class: Hepatotoxicity.
Route of Exposure: Ingestion.
Effects: Chronic ingestion of doses at or slightly above the maximum recommended daily dose can lead to cumulative liver damage. Risk is significantly increased with concurrent alcohol consumption.
Aspiration Hazard: Not classified.
11.2. Other Toxicological Information
Metabolism and Mechanism of Toxicity:
At therapeutic doses, paracetamol is primarily metabolized in the liver via glucuronidation and sulfation to non-toxic metabolites, with a small fraction (5-10%) oxidized by cytochrome P450 (mainly CYP2E1) to a highly reactive, toxic metabolite, N-acetyl-p-benzoquinone imine (NAPQI). Under normal conditions, NAPQI is rapidly detoxified by binding to glutathione.
In overdose, the primary metabolic pathways become saturated, leading to increased metabolism via the P450 pathway. This depletes glutathione stores, allowing NAPQI to accumulate and bind irreversibly to liver cell proteins, causing oxidative stress and cell death (necrosis).
Symptoms of Acute Overdose (Human):
Stage 1 (0.5-24 hours): Nausea, vomiting, sweating, pallor, malaise.
Stage 2 (24-72 hours): Apparent improvement. However, liver enzyme levels (ALT, AST) begin to rise significantly.
Stage 3 (72-96 hours): Peak hepatotoxicity. Symptoms include jaundice (yellowing skin/eyes), confusion (hepatic encephalopathy), coagulopathy, hypoglycemia, and potentially multi-organ failure.
Antidote: N-acetylcysteine (NAC). It acts as a glutathione precursor and substitute, helping to detoxify NAPQI. It is most effective if administered within 8-10 hours of ingestion but is still recommended in later presentations.
Disclaimer: The information presented here is a summary of key toxicological data. It is intended for hazard identification and risk assessment and is not a substitute for a full, supplier-specific Safety Data Sheet or professional medical advice. Always consult the official SDS for comprehensive handling instructions.
From Toxicology to Speculation
Looking at Section 11, the picture is fairly clear: paracetamol is not classified as mutagenic, teratogenic, or carcinogenic. The toxicological red flag is its well-established hepatotoxicity at high doses. That makes sense for how the drug is used clinically—but it also explains where the speculation starts.
Because the mechanisms of overdose are so well described—particularly oxidative stress and glutathione depletion—some researchers have asked whether those same pathways could, in theory, play out in subtler ways during fetal development or early childhood. It’s important to underline that these ideas are extrapolations, not validated conclusions. Still, they form the backbone of the arguments behind a supposed Tylenol–autism link.
With that context, let’s look at the main hypotheses that have been proposed and why they remain unproven.
Based on a comprehensive review of the established toxicological data for paracetamol (acetaminophen), there are no scientifically validated pathways that demonstrate paracetamol causes autism.
The suggestion of a link is a topic of ongoing scientific debate and originates primarily from epidemiological studies that observe correlations, not causation. It is crucial to distinguish between observed associations and proven biological mechanisms.
However, to directly address this question, the proposed hypotheses put forward by researchers who explore this potential link are based on the known toxicology of paracetamol overdose and its extrapolation to potential effects during fetal development or early childhood. These hypotheses remain unproven.
The following flowchart maps out these proposed, but unvalidated, hypothetical pathways:
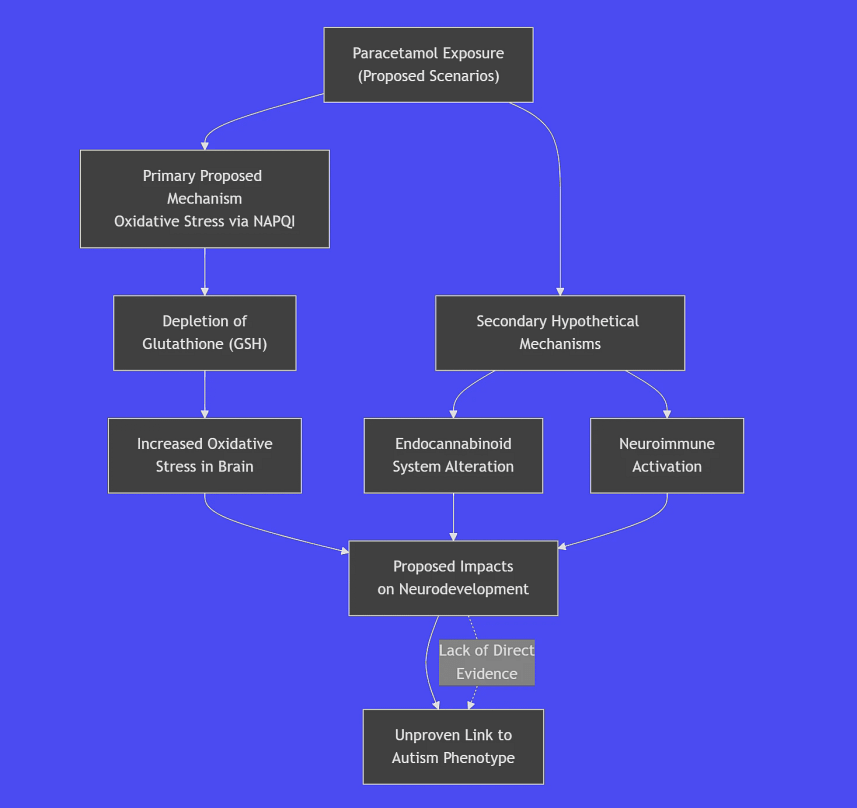

Here is a detailed breakdown of the proposed pathways illustrated above:
1. The Primary Proposed Mechanism: Oxidative Stress and Glutathione Depletion
This is the most frequently cited biological hypothesis, directly derived from the established mechanism of liver toxicity.
The Known Toxicology: As detailed in the SDS, paracetamol overdose depletes the antioxidant glutathione (GSH) in the liver, leading to oxidative stress and cell damage.
The Hypothesis: It is proposed that even therapeutic doses of paracetamol, particularly in a developing fetus (prenatal exposure) or in young infants, could deplete glutathione in the brain. The developing brain is considered highly vulnerable to oxidative stress.
Proposed Chain of Events:
Exposure: A pregnant mother takes paracetamol, or an infant is given paracetamol.
Metabolism: The drug is metabolized, producing low levels of the reactive metabolite NAPQI.
GSH Depletion: It is hypothesized that the immature metabolic systems in the fetus or infant may have a reduced capacity to handle this load, leading to a relative depletion of glutathione in neural tissues.
Oxidative Stress: With reduced antioxidant defenses, the brain experiences increased oxidative stress, which could damage neurons, alter signaling pathways, and trigger neuroinflammation.
Link to Autism: Because autism spectrum disorder (ASD) has been associated with oxidative stress and neuroinflammation in some studies, this pathway is proposed as a potential mechanistic link.
2. Other Hypothetical Pathways
Endocannabinoid System Alteration: Paracetamol metabolism involves the endocannabinoid system (a network of receptors in the brain involved in mood, memory, and neural development). Some researchers hypothesize that paracetamol could alter this system during critical developmental windows, potentially affecting social and emotional brain development.
Neuroimmune Activation: The drug might trigger a mild immune response in the brain (neuroinflammation) in susceptible individuals, which is another area of interest in autism research.
Critical Scientific Context and Major Caveats
It is essential to understand why these pathways remain speculative and are not accepted as causative by major health bodies.
Correlation does not equal Causation: Epidemiological studies finding an association cannot prove that paracetamol causes autism. The association could be explained by confounding factors.
The Most Likely Explanation: The reason for the paracetamol use is often the confounding factor. For example, a pregnant mother taking paracetamol may be doing so for a fever or infection (e.g., flu). It is well-established that high fever and severe infections during pregnancy can themselves influence fetal brain development and are associated with a higher risk of neurodevelopmental disorders. The paracetamol use may simply be a marker for the underlying condition, not the cause.
Lack of Direct Biological Evidence: There is a significant gap between the overdose toxicity seen in the liver and the hypothetical effects on the developing brain at therapeutic doses. No direct, conclusive evidence shows that therapeutic doses of paracetamol cause the proposed oxidative stress or other changes in the human fetal or infant brain.
Position of Major Health Bodies: Organizations like the U.S. Food and Drug Administration (FDA) and the American College of Obstetricians and Gynecologists (ACOG) have reviewed the available evidence and have not found a causal link. They continue to recommend paracetamol as the pain and fever reliever of choice during pregnancy when medication is necessary, as untreated pain and fever can be harmful to the mother and fetus.
In summary, while hypotheses (primarily centered on oxidative stress) have been proposed to suggest a pathway linking paracetamol to autism, these pathways are not supported by conclusive scientific evidence. The observed associations in some studies are more plausibly explained by confounding factors.
The consensus among toxicologists and medical experts is that the known risks of untreated pain and fever during pregnancy outweigh the unproven and highly speculative risk of neurodevelopmental issues from paracetamol use. Always consult a healthcare professional for guidance on medication use during pregnancy or for infants.
The Teratogenicity Question
Before moving deeper into the dose–response debate, it’s important to pause and consider another piece of the toxicological picture: teratogenicity. If paracetamol were truly driving developmental harm, you would expect to see strong evidence of it causing birth defects. But that’s not what the data show.
Across decades of research, paracetamol is not considered a major teratogen. Most large-scale studies have found no consistent link between therapeutic use and structural birth defects. Where signals have appeared—like subtle increases in cryptorchidism or ADHD-like behaviors—the findings are small, inconsistent, and almost certainly clouded by confounding factors such as maternal illness. Unlike well-established teratogens, paracetamol does not interfere directly with embryonic organ formation at therapeutic doses.
This nuance matters, because it sets the stage for the current controversy: the debate isn’t about obvious malformations, but about much subtler and still unproven neurodevelopmental outcomes. Which brings us back to the real crux of the issue—whether incidental, therapeutic use could ever mimic the sustained overdose conditions required to produce known toxic effects.
The Dose Discrepancy
At this point, a reasonable question arises: if the only well-established toxic effect of paracetamol requires a clear overdose, how could incidental, therapeutic use be implicated in something as complex as autism?
That question cuts to the heart of the controversy. The proposed pathways only make sense if you assume a kind of “sustained overdose in disguise”—a scenario where normal use somehow mimics toxic conditions without triggering the obvious, well-documented liver damage. And that assumption is where the theory starts to unravel.
The fundamental and unresolved contradiction is the vast difference between the dose required to cause the known toxic effect (liver damage) and the dose implicated in the unproven association (neurodevelopmental changes).
Let's break down why this dose discrepancy is so problematic for the theory.
1. The Established Toxicity is Dose-Dependent and Has a Clear Threshold
As detailed in the toxicological information:
Mechanism: Toxicity occurs when the primary metabolic pathways (sulfation and glucuronidation) are saturated. This forces the drug down a secondary pathway (CYP2E1 enzyme), producing the toxic metabolite NAPQI.
Saturation Point: This saturation occurs predictably at high doses, typically above the maximum recommended dose of 4 grams per day for adults. The body's defense (glutathione) is overwhelmed, leading to oxidative stress and cell death in the liver.
Key Point: At therapeutic doses, the primary pathways easily handle the metabolism, NAPQI production is minimal, and glutathione readily detoxifies it. No significant oxidative stress occurs systemically.
2. The Hypothesis Requires an Unproven "Vulnerability" Extrapolation
Proponents of the theory must argue that a developing fetus or infant's brain is so uniquely vulnerable that therapeutic doses of paracetamol—which cause no harm to the mother's liver or other organs—somehow produce significant oxidative stress in the fetal brain.
This requires several unsupported leaps:
Leap 1: That the fetal blood-brain barrier and metabolic enzymes are so inefficient that even low, therapeutic levels of paracetamol lead to a problematic accumulation of NAPQI in neural tissue.
Leap 2: That the fetal brain has lower reserves of glutathione or is inherently more susceptible to levels of oxidative stress that are negligible for every other cell in the body.
Leap 3: That this hypothetical, low-grade oxidative stress is sufficient to alter complex neurodevelopmental processes like synaptic pruning and neural migration, but not sufficient to cause more immediate or widespread neurological damage.
In essence, the theory asks us to believe that the fetal brain is simultaneously uniquely vulnerable to the drug's effects, yet also uniquely resilient in showing no other signs of damage except a specific link to autism phenotypes years later. This is a very difficult biochemical case to make.
3. The "Sustained Overdose" vs. "Incidental Use" Distinction
This is where the theory becomes particularly murky and why correlation studies are so unreliable.
Sustained Overdose: If a pregnant woman were to actually overdose on paracetamol, the primary and immediate concern would be acute liver failure and her own death, which would obviously have catastrophic consequences for the fetus. This is a clear, direct toxic effect, but it is not what is being studied or proposed as a cause for autism.
Incidental/Therapeutic Use: The epidemiological studies that suggest a link are based on reported use of paracetamol for headaches, fevers, or pains during pregnancy—in other words, typical, short-term, therapeutic use. There is no evidence that the mothers in these studies were saturating their metabolic pathways.
The Most Parsimonious Explanation
Given the established toxicology, the most scientifically sound position is:
The known mechanism of paracetamol toxicity (oxidative stress via glutathione depletion) is a high-dose phenomenon. There is no validated evidence that this mechanism is activated by incidental, therapeutic use. Therefore, invoking this mechanism to explain a link to autism requires extrapolations that are not supported by fundamental pharmacological principles.
The observed correlations in studies are far more likely explained by confounding factors (the reason for the drug's use, like fever or infection, which themselves can impact neurodevelopment) than by a biologically implausible mechanism that contradicts the drug's well-understood, dose-dependent toxicity profile.
Epidemiology and the Problem of Confounding
And this is where the discussion shifts from toxicology to epidemiology. Because if the dose–response mechanism doesn’t add up, why do some studies still report an association between paracetamol use in pregnancy and later autism diagnoses?
The answer lies less in the chemistry of the drug and more in the messy reality of human health data. Epidemiological studies can detect correlations, but they can’t untangle causation on their own. The most plausible explanation is that the reason for taking paracetamol—most often fever, pain, or infection during pregnancy—is itself the underlying risk factor.
Maternal illness as a driver: High fever and significant infections during pregnancy are well documented to affect fetal brain development. If a mother takes paracetamol for fever, the fever may be the underlying risk, not the medication.
Recall bias and self-reporting: Many of the large studies rely on maternal recall of medication use months or years after pregnancy, which introduces errors and exaggerates weak associations.
Inconsistent findings: Some studies report tiny increases in relative risk, while others show no association at all. When effects are this small and inconsistent, confounding variables are the most plausible explanation.
It’s telling that major health bodies, after reviewing the totality of the evidence, continue to recommend paracetamol as the safest option for pain and fever relief in pregnancy. The potential harms of untreated fever—miscarriage, preterm birth, or serious neurodevelopmental risk—are concrete and immediate, whereas the autism hypothesis remains speculative.
In short: the epidemiological data raise interesting questions, but they don’t override the toxicology, nor do they establish causation.
The paracetamol–autism controversy sits at the intersection of weak correlations, biological speculation, and the amplification of uncertainty by public figures and media.
While epidemiology raises questions, it can’t explain why only some children in these studies develop autism while the vast majority—despite similar exposures—do not. To answer that, we need to zoom out from chemistry and population statistics and look at genetics.
Autism has one of the highest heritability estimates of any neurodevelopmental condition, which means genetic predisposition is the dominant factor. The role of environmental exposures like fever—or, hypothetically, paracetamol—makes sense only when understood through this lens of genetic vulnerability. This is where the “two-hit” hypothesis becomes a useful model.
There is a crucial question that gets to the heart of the "trigger vs. cause" distinction and the complex interplay between genetics and environment in autism spectrum disorder (ASD) - what role does genetic predisposition play.
Here’s a breakdown of how the predisposition (heritability) relates to potential environmental triggers like fever or the hypothetical role of paracetamol.
The Core Concept: The Two-Hit Hypothesis
A useful model for understanding this is the "two-hit" hypothesis or "gene-environment interaction" model.
Hit 1 (Genetic Predisposition): An individual is born with a certain set of genetic variants that increase their susceptibility or vulnerability to developmental disruptions. In the case of autism, this is well-established—heritability is very high, estimated at ~80%. This doesn't mean 80% of cases are purely genetic, but that genetic differences account for about 80% of the variation in risk within a population.
Hit 2 (Environmental Trigger): An environmental factor (like a severe fever, infection, or hypothetically a toxin) occurs during a critical window of brain development (prenatally or early postnatal). This factor may not cause problems in a typically developing brain but can "trigger" a cascade of abnormal development in a genetically vulnerable brain.
Applying the Model to the Question
Scenario 1: High Fevers as a Trigger
This is a plausible and actively researched trigger.
The Mechanism: A high fever represents a significant biological stressor. It can activate the mother's (if during pregnancy) or the infant's immune system, leading to the release of inflammatory molecules called cytokines. These cytokines can cross the placenta or the blood-brain barrier and can interfere with key neurodevelopmental processes like neuron migration and synapse formation.
Interaction with Genetics: A brain with a genetic predisposition to autism might be particularly sensitive to this inflammatory insult. The same fever that causes no lasting issues in a neurotypical child might disrupt development in a vulnerable child, acting as the "second hit" that contributes to the autism phenotype.
Crucially, this makes the fever (or the underlying infection) the potential trigger, not the paracetamol. In fact, this is a primary argument against the paracetamol theory: failing to treat a serious fever could pose a greater neurodevelopmental risk than the medication itself.
Scenario 2: Paracetamol as a Trigger (The Unproven Hypothesis)
If paracetamol were a trigger (which remains unproven), the model would apply similarly.
The Proposed Mechanism: In a genetically vulnerable fetus/child, the ability to metabolize paracetamol without oxidative stress might be compromised. For example, they might have naturally lower levels of glutathione or more active CYP enzymes. In this case, even a standard therapeutic dose could theoretically lead to higher-than-expected oxidative stress in the brain during a critical developmental period.
Key Point: In this model, the drug would have no effect on a child without this genetic predisposition. The effect is entirely dependent on the interaction between the environmental exposure (the drug) and the genetic background (the vulnerability).
Applicability Irrespective of Genetic Position?
The answer is no, the trigger is not applicable irrespective of genetics. The entire concept of a "trigger" implies a pre-existing vulnerability.
For a child with no genetic predisposition: A high fever or a dose of paracetamol is highly unlikely to cause autism. Their neurodevelopmental systems are resilient enough to handle the insult without being pushed off a typical trajectory.
For a child with a high genetic load: Their development may be on a "knife's edge." A significant environmental insult (like a severe infection/fever) during a key window could be the factor that contributes to the manifestation of ASD.
In conclusion: The heritability of autism suggests that for most individuals, a genetic predisposition is the foundational "Hit 1." Environmental factors like high fevers are best understood as potential "Hit 2" triggers that can modulate risk, but their effect is profoundly influenced by the underlying genetic landscape. This explains why the vast majority of children who experience fevers or take paracetamol do not develop autism.
This framing matters, because it explains why the paracetamol hypothesis feels shaky: it ignores the genetic baseline. A trigger can only act on a predisposition. For most children, fevers or paracetamol use during development won’t push anything off course. For those with a high genetic load, the underlying risk is already present, and environmental stressors may only modulate—not cause—the outcome.
Which brings us back to the real-world question: why, despite weak science, does the Tylenol–autism claim continue to circulate so powerfully in public debate?
Why the Debate Persists
This is where science and public discourse part ways. Toxicology tells us that therapeutic paracetamol doesn’t reach the thresholds needed for systemic harm. Epidemiology, at best, shows weak and inconsistent correlations that can’t separate the drug from the underlying conditions it treats. Yet in the gap between “no proven mechanism” and “some statistical noise,” speculation flourishes.
This is why the controversy persists: because uncertainty is easy to amplify, while nuance is harder to communicate. Public figures and media outlets know that “Tylenol causes autism” grabs attention in a way that “weak associations confounded by fever” never will.
So the real task isn’t just to parse toxicology tables or critique study design—it’s to equip ourselves and others with the tools to tell solid evidence from shaky conjecture. That means returning to the source data, questioning the leaps made between correlation and causation, and resisting the lure of simplified narratives when the science is anything but simple.
